Onapristone
 | |
| Clinical data | |
|---|---|
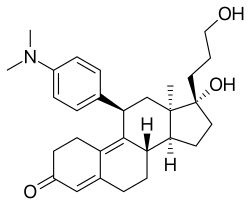
| Other names | ZK-89299; ZK-299; AR-18; IVV-1001; 11β-(4-(Dimethylamino)phenyl)-17α-hydroxy-17β-(3-hydroxypropyl)-13α-estra-4,9-dien-3-one |
| Drug class | Antiprogestogen |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| ECHA InfoCard | 100.233.493 |
| Chemical and physical data | |
| Formula | C29H39NO3 |
| Molar mass | 449.635 g·mol−1 |
| 3D model (JSmol) |
|
| |
InChI
| |
Onapristone (INNTooltip International Nonproprietary Name) (developmental code names ZK-89299, ZK-299) is a synthetic and steroidal antiprogestogen with additional antiglucocorticoid activity which was developed by Schering[1] and described in 1984 but was never marketed.[2][3] It is a silent antagonist of the progesterone receptor (PR), in contrast to the related antiprogestogen mifepristone (which is a weak partial agonist of the receptor).[4] Moreover, compared to mifepristone, onapristone has reduced antiglucocorticoid activity, shows little antiandrogenic activity, and has 10- to 30-fold greater potency as an antiprogestogen.[4] The medication was under development for clinical use, for instance in the treatment of breast cancer and as an endometrial contraceptive, but was discontinued during phase III clinical trials in 1995 due to findings that liver function abnormalities developed in a majority patients.[5][6][7]
Onapristone has been found to be effective in the treatment of breast cancer.[8][5][9]
As of 2016, onapristone has re-emerged and is under development for the treatment of prostate cancer, currently in phase II clinical trials.[10] It was also under development for the treatment of endometrial cancer, breast cancer, ovarian cancer, and uterine cancer, but was discontinued for these indications in favor of focusing on prostate cancer.[10]
See also
- List of investigational sex-hormonal agents § Progestogenics
- Aglepristone
- Lilopristone
- Telapristone
- Toripristone
References
- ^ Lange CA, Sartorius CA, Abdel-Hafiz H, Spillman MA, Horwitz KB, Jacobsen BM (2008). "Progesterone receptor action: translating studies in breast cancer models to clinical insights". Advances in Experimental Medicine and Biology. Vol. 630. Springer. pp. 94–111. doi:10.1007/978-0-387-78818-0_7. ISBN 978-0-387-78817-3. PMID 18637487. Onapristone, p. 102, at Google Books
- ^ Elks J, Ganellin CR (1990). "O". Dictionary of Drugs. Springer. pp. 892–927. doi:10.1007/978-1-4757-2085-3_15. ISBN 978-1-4757-2087-7. Onapristone, p. 903, at Google Books
- ^ Morton IK, Hall JM (1999). "O". Concise Dictionary of Pharmacological Agents. Springer. pp. 206–213. doi:10.1007/978-94-011-4439-1_14. ISBN 978-94-010-5907-7. Onapristone, p. 207, at Google Books
- ^ a b Pavlik EJ, Nelson K, Srinivasan S, Depriest PD, Kenady DE (1997). "Antiestrogen Resistance in Human Breast Cancer". Estrogens, Progestins, and Their Antagonists. Hormones in Health and Disease. Birkhäuser. pp. 115–160. doi:10.1007/978-1-4612-4096-9_5. ISBN 978-1-4612-8650-9. Onapristone, p. 134, at Google Books
- ^ a b Robertson JF, Willsher PC, Winterbottom L, Blamey RW, Thorpe S (February 1999). "Onapristone, a progesterone receptor antagonist, as first-line therapy in primary breast cancer". European Journal of Cancer. 35 (2): 214–218. doi:10.1016/S0959-8049(98)00388-8. PMID 10448262.
- ^ Katkam RR, Gopalkrishnan K, Chwalisz K, Schillinger E, Puri CP (September 1995). "Onapristone (ZK 98.299): a potential antiprogestin for endometrial contraception". American Journal of Obstetrics and Gynecology. 173 (3 Pt 1): 779–787. doi:10.1016/0002-9378(95)90341-0. PMID 7573244.
- ^ Howell SJ, Howell A (2010). "Endocrine Therapy". Management of Breast Diseases. Springer. pp. 329–352. doi:10.1007/978-3-540-69743-5_18. ISBN 978-3-540-69742-8. Onapristone, p. 338, at Google Books
- ^ Klijn JG, Setyono-Han B, Foekens JA (2000). "Progesterone antagonists and progesterone receptor modulators in the treatment of breast cancer". Steroids. 65 (10–11): 825–830. doi:10.1016/S0039-128X(00)00195-1. PMID 11108894. S2CID 25524094.
- ^ Cottu PH, Bonneterre J, Varga A, Campone M, Leary A, Floquet A, et al. (2018). "Phase I study of onapristone, a type I antiprogestin, in female patients with previously treated recurrent or metastatic progesterone receptor-expressing cancers". PLOS ONE. 13 (10): e0204973. Bibcode:2018PLoSO..1304973C. doi:10.1371/journal.pone.0204973. PMC 6179222. PMID 30304013.
- ^ a b "Onapristone - Context Therapeutics". Adis Insight. Springer Nature Switzerland AG.
- v
- t
- e
- See also
- Receptor/signaling modulators
- Glucocorticoids and antiglucocorticoids
- Mineralocorticoid receptor modulators
- List of corticosteroids











