Truxene
 | |
| Names | |
|---|---|
| Preferred IUPAC name 10,15-dihydro-5H-diindeno-[1,2-a:1',2'-c]fluorene | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.008.132 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C27H18 |
| Molar mass | 342.441 g·mol−1 |
| Density | 1.286 g/cm3 |
| Melting point | 378 °C (712 °F; 651 K) [1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
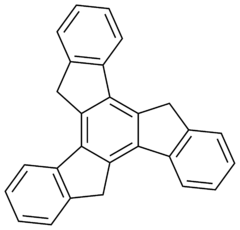
Truxene is a polycyclic aromatic hydrocarbon. The molecule can be thought of as being made up of three fluorene units arranged symmetrically and sharing a common central benzene. Truxene is solid, and it is slightly soluble in water.
History
Truxene has been known since the end of the 19th century. J. Hausmann came across it in 1889 while investigating the reactions of 3-phenylpropionic acid with phosphorus pentoxide. He could not determine the exact structure but assumed it was a cyclic trimer of 1-indanone. According to him, it was formed by the condensation of 1-indanone resulting from intramolecular acylation of 3-phenylpropanoic acid.[2]
Frederic Stanley Kipping was able to confirm the structure of truxene in 1894 and obtained the compound by the trimerization of 1-indanone.[3]
Preparation
Truxene is prepared by the cyclotrimerization of 1-indanone in a mixture of acetic acid and concentrated hydrochloric acid. [4]
Uses
Truxene has a star shape, and it is therefore suitable as a starting point for the synthesis of dendrimers.[5]
Derivatives of truxene have also been used for the synthesis of liquid crystals[6] and fragments of fullerene.[7]
References
- ^ Harper, William L.; Smith, Wesley E. Process for synthesizing truxene; amorphous or graphitic carbon from indenes. 1970. US 3504044 A.
- ^ Hausmann, J. (July 1889). "Einwirkung von o -Cyanbenzylchlorid auf Natriummalonester. Untersuchung des α-Hydrindons". Berichte der Deutschen Chemischen Gesellschaft. 22 (2): 2019–2026. doi:10.1002/cber.18890220264.
- ^ Kipping, F. Stanley (1894). "XXIX. The formation of the hydrocarbon "truxene" from phenylpropionic acid, and from hydrindone". J. Chem. Soc., Trans. 65: 269–290. doi:10.1039/CT8946500269.
- ^ Amick, Aaron Warren (2008). Methodology Development for Use in Polycyclic Aromatic Hydrocarbon Synthesis. PhD thesis. p. 5. ISBN 978-0-549-75717-7.
- ^ Cao, Xiao-Yu; Zhang, Wen-Bin; Wang, Jin-Liang; Zhou, Xing-Hua; Lu, Hua; Pei, Jian (1 October 2003). "Extended π-Conjugated Dendrimers Based on Truxene". Journal of the American Chemical Society. 125 (41): 12430–12431. doi:10.1021/ja037723d. PMID 14531685.
- ^ Destrade, C.; Gasparoux, H.; Babeau, A.; Tinh, Nguyen Huu; Malthete, J. (May 1981). "Truxene Derivatives: A New Family of Disc-Like Liquid Crystals With an Inverted Nematic-Columnar Sequence". Molecular Crystals and Liquid Crystals. 67 (1): 37–47. doi:10.1080/00268948108070873.
- ^ Dehmlow, Eckehard V.; Kelle, Torsten (June 1997). "Synthesis of New Truxene Derivatives: Possible Precursors of Fullerene Partial Structures?". Synthetic Communications. 27 (11): 2021–2031. doi:10.1080/00397919708006804.
- v
- t
- e
- Benz[a]anthracene
- Benzo[a]fluorene
- Benzo[c]fluorene
- Benzo[c]phenanthrene
- Chrysene
- Fluoranthene
- Pyrene
- Tetracene
- Triphenylene
- Tricyclobutabenzene
- Benz[e]acephenanthrylene
- Benzopyrene
- Benzo[a]pyrene
- Benzo[e]pyrene
- 6H-Benzo[cd]pyrene(Olympicene)
- Benzo[a]fluoranthene
- Benzo[b]fluoranthene
- Benzo[j]fluoranthene
- Benzo[k]fluoranthene
- trans-Bicalicene
- Dibenz[a,h]anthracene
- Dibenz[a,j]anthracene
- Pentacene
- Perylene
- Picene
- Tetraphenylene
- Anthanthrene
- Benzo[ghi]perylene
- Corannulene
- Dibenzopyrenes
- Hexacene
- Triangulene
- Zethrene












